Welcome back to Healthy Innovations! 👋
Healthy Innovations is the newsletter for forward-looking healthcare leaders who want to learn about the latest advances in this fast-paced industry. From AI-powered diagnostics to revolutionary gene therapies, I will highlight the fascinating breakthroughs reshaping healthcare and explain what this means for you, your business, and the wider community.
In this issue of Healthy Innovations, we are deep diving into the world of CRISPR technology. According to a report by the Future Today Institute, the size of the global market for CRISPR technologies is projected to exceed $4 billion by mid-decade. In the US alone, CRISPR could contribute $19 billion to GDP by 2032.
So, let's explore how this revolutionary biotechnology is reshaping medicine as we know it.
Remember when CRISPR was just a fascinating lab technique? Well, 2024 marked the year this revolutionary gene-editing tool finally entered mainstream medicine, and 2025 is shaping up to be even more transformative.
Let's explore where we are and what's coming next.
What is CRISPR?
CRISPR is an acronym for Clustered Regularly Interspaced Short Palindromic Repeats, a family of DNA sequences found in the genomes of prokaryotic organisms like bacteria.
These sequences are part of a microbial immune system that detects and destroys viral DNA during infections, providing a form of acquired immunity. In biotechnology, CRISPR is widely known as the foundation for the CRISPR-Cas9 genome editing system, which allows scientists to modify DNA precisely.
The system includes two main components: the Cas9 protein, which acts as molecular scissors to cut DNA, and a guide RNA (gRNA) that directs Cas9 to specific genetic sequences.
Over the last 10 years, CRISPR-Cas9-based treatments have been tested in various conditions, including cancers, inherited diseases, HIV and diabetes. However, the most significant progress has been seen in blood disorders.
🥇 The development of CRISPR-Cas9 technology earned Emmanuelle Charpentier and Jennifer Doudna the 2020 Nobel Prize in Chemistry for its transformative impact on science and medicine
The first step: Casgevy makes history
The 2023 approval of Casgevy (exagamglogene autotemcel) by both FDA and UK regulators marked a watershed moment in medical history.
This first CRISPR-Cas9-based therapy from Vertex targets sickle cell disease (SCD) and transfusion-dependent beta-thalassemia (TDT) by editing the BCL11A gene to trigger the production of fetal hemoglobin, effectively treating these devastating blood disorders.
Data presented at ASH 2024 showed that:
In SCD, 39/42 (93%) evaluable patients (those with at least 16 months of follow-up) were free from vaso-occlusive crises (VOCs) - primary cause of severe pain and emergency hospitalizations in patients - for at least 12 consecutive months.
In TDT, 53/54 (98%) evaluable patients (those with at least 16 months of follow-up) achieved transfusion-independence for at least 12 consecutive months.
Both SCD and TDT patients reported sustained and clinically meaningful improvements in their quality of life, including physical, emotional, social/family and functional well-being, and overall health status.
Casgevy is designed as a one-time gene therapy with potentially life-long benefits and while the treatment's effectiveness is remarkable, its $2 million price tag raises crucial questions about accessibility. The process involves complex procedures: extracting blood-producing stem cells, precisely editing them, and carefully reintegrating them into patients. The treatment also requires patients undergo multiple blood transfusions, and a round of chemotherapy before getting the cells, and a month in the hospital afterward.
Given the technological sophistication, high cost, and demands on patients, initial uptake of the CRISPR treatment has been slow. However, clinicians are optimistic that demand will increase as more success stories emerge.
Beyond the cut: Next-generation gene editing
The original CRISPR-Cas9 system was just the beginning.
Today's genetic medicine toolkit has expanded dramatically with more precise and versatile approaches:
Base editing, developed by Harvard's David Liu, enables the alteration of individual DNA letters without cutting the double helix. Beam Therapeutics is already conducting clinical trials for leukemia treatment using this technology, while Verve Therapeutics shows promising results in treating high cholesterol.
Prime editing offers even greater precision, allowing scientists to delete or add small DNA segments with fewer off-target effects than traditional CRISPR. Think of it as a genetic "search-and-replace" function, offering unprecedented control over DNA modifications.
Perhaps most intriguingly, epi-editing has emerged as a way to regulate gene expression without altering the underlying DNA. Companies like Tune Therapeutics and Epic Bio are preparing to test this approach in clinical trials, potentially offering new treatments for previously untreatable conditions.
The in vivo revolution
One of the most exciting developments is the ability to perform gene editing directly inside the body.
Intellia Therapeutics' NTLA-2001 represents a breakthrough in this area - a single intravenous treatment for transthyretin amyloidosis (a rare, progressive and fatal disease) that targets liver cells to disable disease-causing genes. Early trials have shown remarkable results, with up to 93% reduction in problematic proteins, maintaining stability for at least nine months.
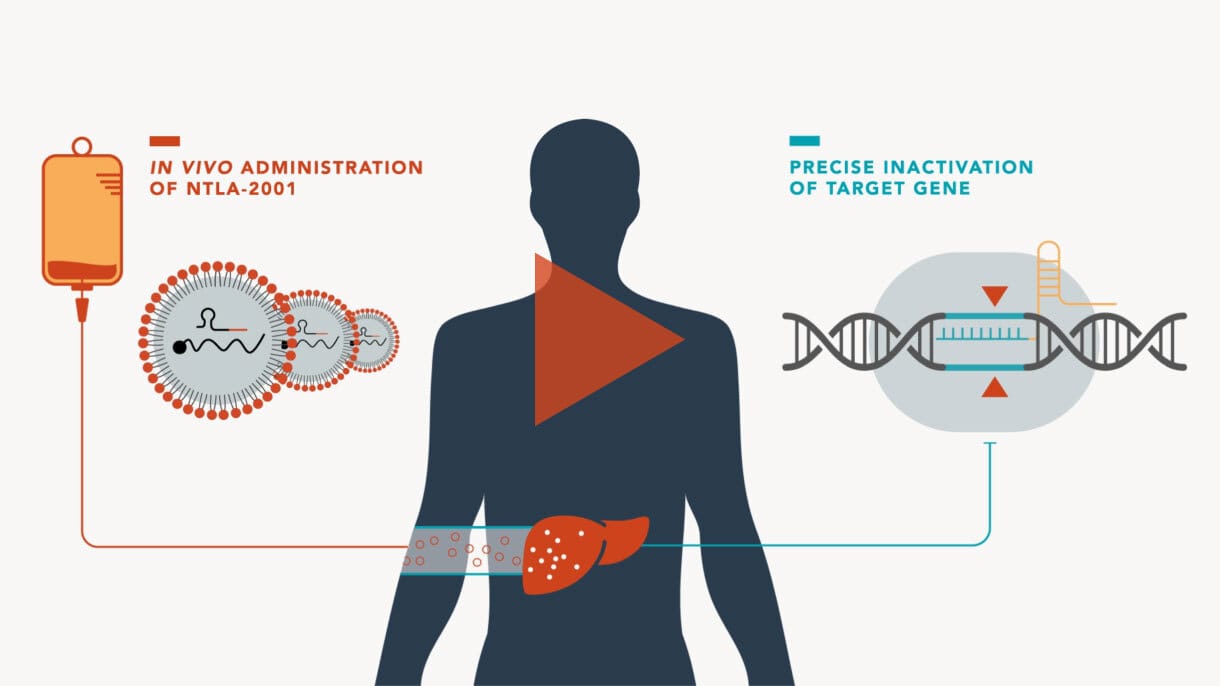
Image from intelliatx.com
This shift from laboratory manipulation to direct body treatment could revolutionise how we approach genetic diseases. Imagine treating cancer not with traditional chemotherapy but with precisely targeted genetic modifications. However, this advancement comes with important considerations about off-target effects and potential impacts on reproductive cells.
🔮 Looking ahead
As we move further into 2025, the convergence of CRISPR with AI promises to accelerate discovery and improve precision. The market reflects this potential, growing from $3.12 billion in 2022 to $4.69 billion in 2024. Yet challenges remain, particularly in making these transformative treatments more accessible.
The question is no longer whether CRISPR will transform medicine and biotechnology but how quickly these transformations will reach everyday practice. As costs decrease and techniques improve, we may be looking at a future where genetic medicine is as common as bone marrow transplants are today.
What do you think about these developments? Are we moving too fast or not fast enough?
📚 Reading recommendation: If you're interested in learning more about Jennifer Doudna and the science of CRISPR, I highly recommend checking out The Code Breaker by Walter Isaacson.
Innovation highlights
🤖 AI decodes diabetes: A study has demonstrated GPT-4's ability to interpret continuous glucose monitoring (CGM) data with remarkable accuracy. The AI system successfully analyzed 14-day glucose patterns and generated clinical summaries that received high marks from healthcare professionals. While some refinements are still needed, the technology achieved 90% accuracy in metric calculations, potentially transforming how patients and clinicians interpret and act on CGM data.
⚛️ Brain cancer's new nemesis: Mayo Clinic researchers have achieved a breakthrough in treating glioblastoma, the most aggressive form of brain cancer, by combining advanced imaging (18F-DOPA PET) with targeted proton beam therapy. The novel approach extended median survival from 6-9 months to over 13 months in patients over 65, with some living beyond two years. This shorter, more effective treatment protocol not only improves survival rates but also reduces the burden on patients by cutting treatment duration from 6 weeks to just 1-2 weeks.
👁️ Molecules a sight for sore eyes: New compounds show promise for treating retinitis pigmentosa, affecting 100,000 Americans. Using virtual screening, researchers found molecules that stabilize rhodopsin protein across 36 genetic subtypes, protecting against retinal degeneration in mice. These molecules can safely cross blood-retina barriers, offering potential life-changing treatment for progressive vision loss.
Cool tool
🔍📑 Looking to stay on top of scientific research without drowning in papers? Consensus AI might be your new best friend. This clever academic search engine is like having a team of research assistants at your fingertips. Simply ask a question in plain English, and it will analyze over 200 million peer-reviewed papers to give you evidence-backed answers and summaries.
What makes it special? Its "Consensus Meter" shows you when researchers agree (or disagree) on a topic, making it perfect for both quick fact-checks and deep research dives. While the free version is great for casual use, power users might want to consider the paid tier for unlimited searches and advanced features.
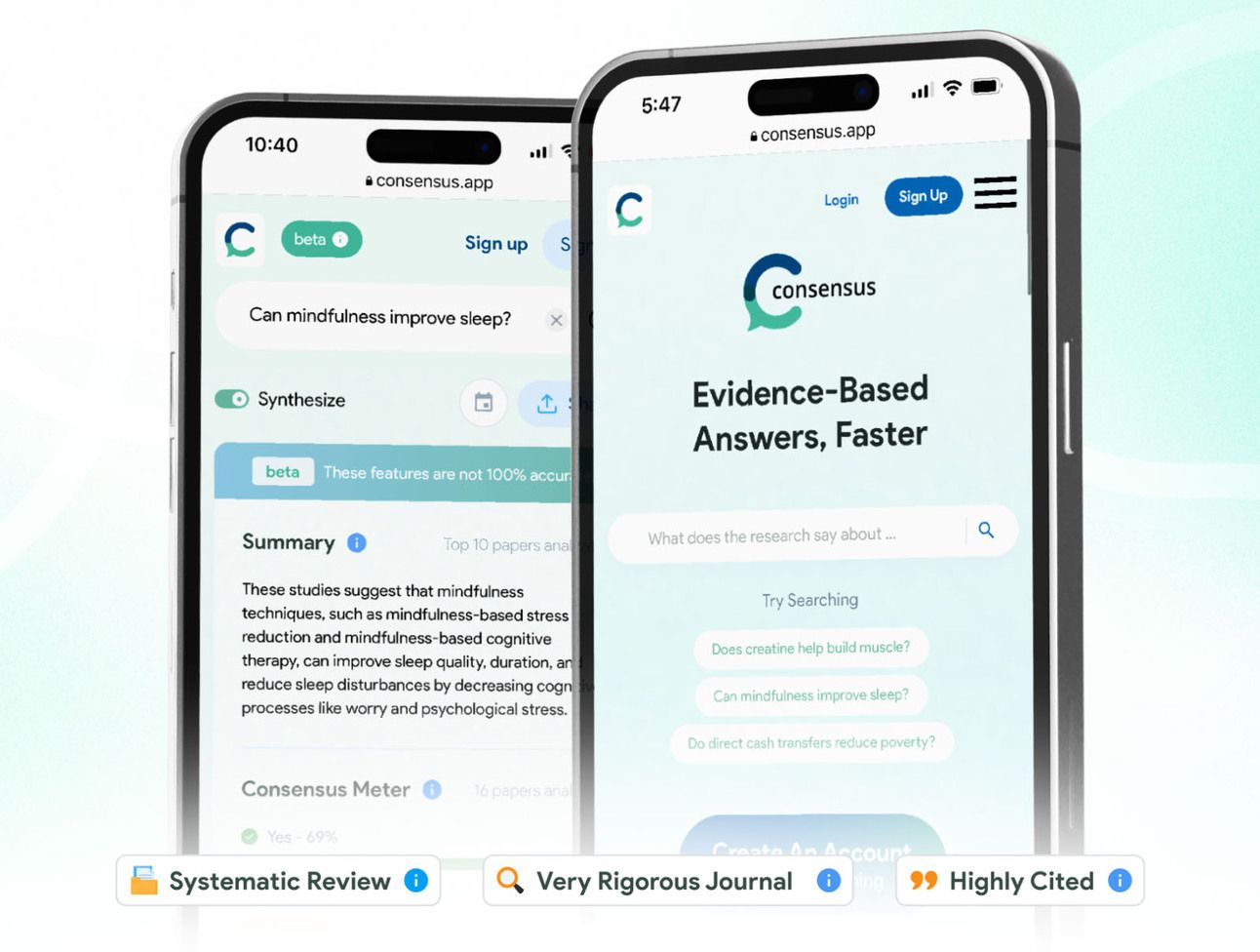
Image from Elastic.co
Company to watch
🧪 Grove AI is targeting clinical trials with its innovative AI-powered platform. Founded by Stanford researchers Tran Le and Sohit Gatiganti, the company's virtual assistant, Grace, handles everything from participant prescreening to appointment scheduling, reducing enrollment times from weeks to minutes. Having supported over 70,000 participants with a 97% satisfaction rate in its first year and fresh off a $4.9 million seed round led by A* Ventures, Grove AI is well-positioned to transform the $10 billion clinical trial management market. Early adopters already report significant improvements in their Alzheimer's and vaccine trials, suggesting this could be a game-changer for clinical research efficiency.

Weird and wonderful
💩 Poop pills for diabetes: A recent clinical trial has shown that fecal microbiota transplantation (FMT) capsules could be a game-changer for type 1 diabetes patients. The study found that patients taking these specialized pills containing donated gut microbiomes saw their gastrointestinal symptom severity scores drop significantly (42 to 25) compared to placebo. While the trial was small, it marks one of the most successful applications of FMT beyond treating C. difficile infections, potentially opening new doors for diabetes care management.
Thank you for reading the Healthy Innovations newsletter!
Keep an eye out for next week’s issue, where I will highlight the healthcare innovations you need to know about.
Have a great week! Alison ✨
P.S. If you enjoyed reading the Healthy Innovations newsletter, please subscribe so I know the content is valuable to you!
P.P.S. Check out beehiiv (affiliate link) if one of your 2025 resolutions is to start your very own newsletter.


